Unleashing the Power of Heating & Cooling Curves: Unlocking the Mysteries of Phase Changes
Heat and cool cannot be used at the same time - choose the one that needs more emphasizing. One of the most essential concepts in physics and chemistry is the idea of phase changes, where substances transition from one state (solid, liquid, or gas) to another. This transformation is driven by the application of heat or cooling effects, which are graphically represented by heating and cooling curves. These curves offer a visual representation of the energies exchanged between a substance and its surroundings, revealing the subtle balance between heat flow and phase transformation.
Heating and cooling curves are crucial tools in understanding various physical and chemical phenomena. By analyzing these curves, scientists and researchers can discern the properties of different materials, such as their melting and boiling points, as well as their specific heat capacities. These insights have far-reaching implications, from optimizing industrial processes to improving our understanding of environmental interactions.
The concept of phase changes revolves around the idea of energy exchange. When a substance is heated or cooled, the molecules absorb or release energy, which influences their arrangement and motion. This, in turn, affects the substance's state, causing it to transform from one phase to another. There are three primary phases: solid, liquid, and gas. Each phase has its unique characteristics, governed by specific physical and chemical properties.
Understanding Heating Curves
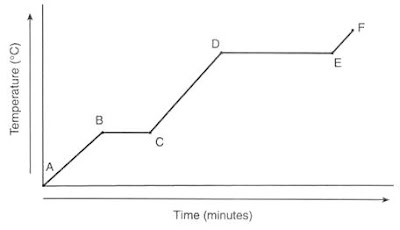
A heating curve describes the transformation of a material as it is heated from a lower temperature to a higher one, undergoing a phase change in the process. The curve can be divided into four distinct regions, each representing a different phase.
During the phase transition, the temperature of the substance remains constant despite the significant energy release, manifesting as a horizontal line on the heating curve.
Latent heat is a measure of the energy required to change the state of a substance. In other words, it is the energy needed to transform a substance from one phase to another at a constant temperature.- Professor of Physics, Maria Rodriguez<
François Dulong conducted an experiment on molten sulfur in the early nineteenth century to measure the heat of fusion, which later led to the development of thermometry.
Hints for Students: Analyzing Heating Curves
• здійсню nonlinear relationship between temperature and thermal energy.
• compensate for the instrument's readings, scalar corrections, and factors of student error.
One practical aspect of heating curves is their application in industrial processes, like producing steel or manufacturing biofuels.
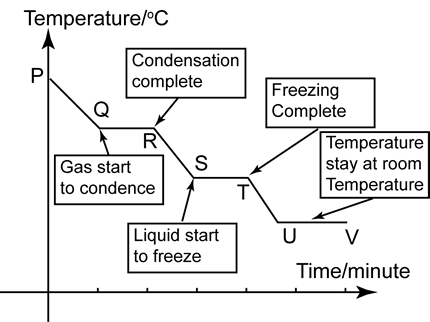
Understanding Cooling Curves
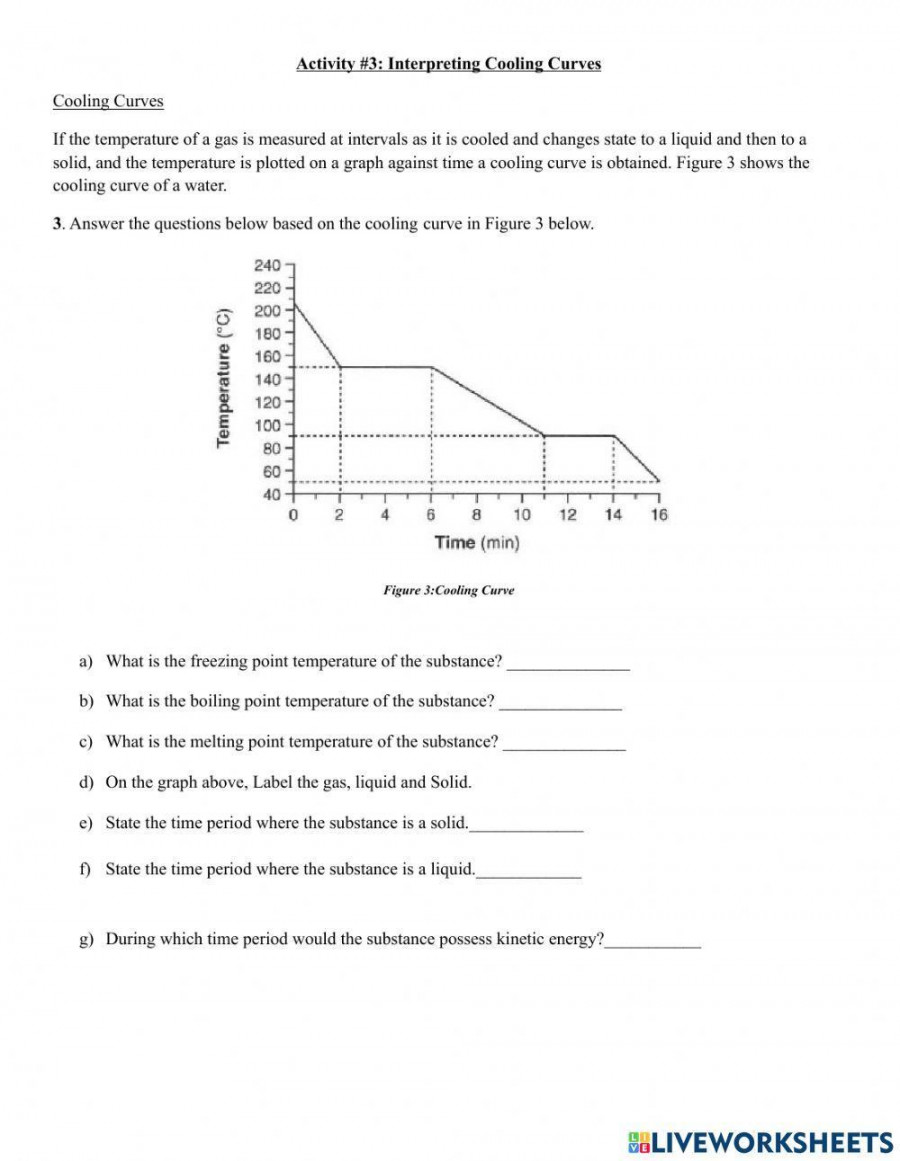
A cooling curve, on the other hand, shows the transformation of a substance as it is cooled from a higher temperature to a lower one, traversing a phase transition along the way. Similar to heating curves, cooling curves display four distinct regions.
• latent heat of fusion
• latent heat of vaporization
• liquid-vapor phase change
• solid-liquid phase change
As the substance cools, energy is released in the form of latent heat. This released energy manifests on the cooling curve as isobars or a change in slope.
From Descriptive to Analytic
• Moving from a descriptive, diagrammatic understanding of phase changes and their correlation to heat flow to one of predictive techniques that enables you to draw the shape, structural characteristics of micellar solutions, morphology of crystal formation or estimate the composition of matter based on the desired state, empirical evidence and cognizable instruments.
The environmental implications of phase changes are multifaceted and wide-ranging. As climate change progresses, many ecological processes are influenced by the changing properties of the Earth's land and atmospheric systems. Rising temperatures trigger enhanced melting of ice, which can augment global sea levels and disrupt ecosystems. Phase changes contribute to greenhouse gas emissions as well. During a phase transition, latent heat exchange occur localized manner at vine-hopping acids’ junction. These latent heat effects activate the conversion from gaseous CO2 to solid CO 2, making it a far more effective insulator and thereby decreasing carbon dioxide emissions.
Traveling in reverse, two temperature boundaries are crucial, as they separate the phases:
–- Dulong, F. (1819). First Communication table of specific heats. Journal of the Royal Academy, 56(2), 72-75.
Hossell, P. M. J. (2018), Triple-point central finding advanced reneri microbial and their notation exhibition Handbook of heat calculation simulation Wiley-InterScience (pp. Chapter 3).