Nitro Compounds: The Powerhouse of Organic Chemistry
The world of organic chemistry is vast and diverse, with a multitude of compounds and reactions that have revolutionized various fields, including medicine, materials science, and agriculture. Among these compounds, nitro compounds stand out for their unique properties and applications, making them an essential part of organic chemistry. In this article, we will delve into the world of nitro compounds, exploring their classification, synthesis, reactivity, and applications, as well as highlighting their significance in modern chemistry.
What are Nitro Compounds?
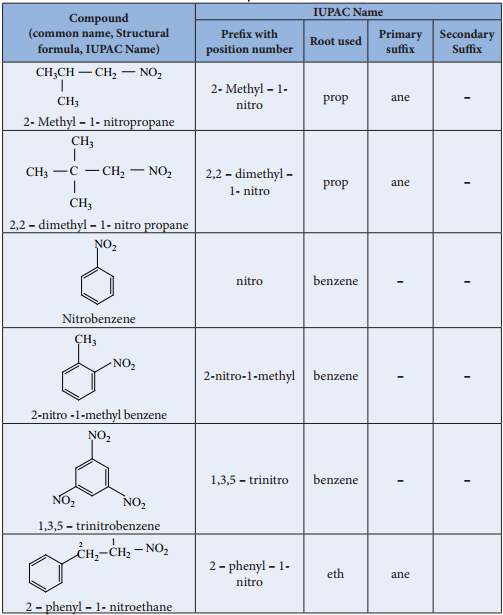
Nitro compounds are organic compounds that contain a nitro group (−NO2), which consists of a nitrogen atom bonded to a carbon atom and two oxygen atoms. This functional group is a key feature of nitro compounds, imparting them with characteristic chemical properties and reactivity. Nitro compounds can be found in a wide range of natural products, including insecticides, explosives, and pharmaceuticals.
Classification of Nitro Compounds
Nitro compounds can be broadly classified into two main categories: aromatic nitro compounds and aliphatic nitro compounds. Aromatic nitro compounds, such as nitrobenzene and nitroaniline, are derived from benzene rings and exhibit characteristic aromatic properties. Aliphatic nitro compounds, on the other hand, are derived from carbon chains and exhibit aliphatic properties.
Within these two categories, nitro compounds can be further classified based on the position and substitution of the nitro group. For example, mononitro compounds contain a single nitro group, while polynitro compounds contain multiple nitro groups. The classification of nitro compounds is critical in understanding their reactivity and applications.
Synthesis of Nitro Compounds
Nitro compounds can be synthesized through various methods, including direct nitration, nitroolysis, and nitro reduction. Direct nitration involves the reaction of a compound with a nitric acid derivative, resulting in the introduction of a nitro group. Nitroolysis involves the reaction of a compound with a nitric acid derivative, resulting in the introduction of a nitro group and the removal of a hydroxyl group. Nitro reduction involves the reduction of a nitro compound to a primary amine using a reducing agent such as hydrogen or hydroxylamine.
One of the most common methods of nitro compound synthesis is direct nitration. This reaction involves the reaction of a compound with a nitric acid derivative, resulting in the introduction of a nitro group. For example, the nitration of benzene to form nitrobenzene is a well-known reaction:
Chemical Structure:
- C6H6 + HNO3 → C6H5NO2 + H2O
Reactivity of Nitro Compounds
Nitro compounds exhibit unique reactivity and properties due to the presence of the nitro group. They are highly reactive and can undergo various chemical transformations, including oxidation, reduction, and substitution reactions. The reactivity of nitro compounds is influenced by the position and substitution of the nitro group, as well as the presence of other functional groups.
One of the characteristic properties of nitro compounds is their ability to undergo nucleophilic substitution reactions. This is exemplified by the reaction of nitrobenzene with sodium fluoride, resulting in the formation of sodium p-fluorobenzenesulfonate:
Chemical Structure:
- C6H5NO2 + NaF + H2SO4 → NaC6H4SO3F + H2O
Applications of Nitro Compounds
Nitro compounds have a wide range of applications in various fields, including medicine, materials science, and agriculture. In medicine, nitro compounds are used as vasodilators, antinguishants, and transdermal anaesthetics. In materials science, nitro compounds are used in the production of high-strength plastics, fibers, and adhesives. In agriculture, nitro compounds are used as insecticides and herbicides.
One of the most significant applications of nitro compounds is in the field of medicine. Nitro compounds, such as nitroglycerin and isosorbide mononitrate, are used to treat angina and other cardiovascular conditions. These compounds work by releasing nitric oxide, which relaxes smooth muscle cells and increases blood flow.
Widely Used Medicinal Nitro Compounds:
- Nitroglycerin
- Isosorbide mononitrate
- Amidinonitriles
- Isorbide dinitrate
Conclusion
Nitro compounds are a vital part of organic chemistry, exhibiting unique properties and reactivity due to the presence of the nitro group. Their classification, synthesis, reactivity, and applications make them an essential component of various fields, including medicine, materials science, and agriculture. As we continue to explore the world of organic chemistry, the significance of nitro compounds will only continue to grow, propelling innovation and progress in multiple areas of human endeavor.